Two different mechanisms of stabilization of regular p-stacks of radicals in switchable dithiazolyl-based materials.
T. Francese, S. Vela, M. Deumal, F. Mota, J.J. Novoa, M. Farnesi Camellone, S. Fabris, R.W.A. Havenith, R. Broer, J. Ribas-Arino.
J. Mater. Chem. C., 8 (2020) 5437.
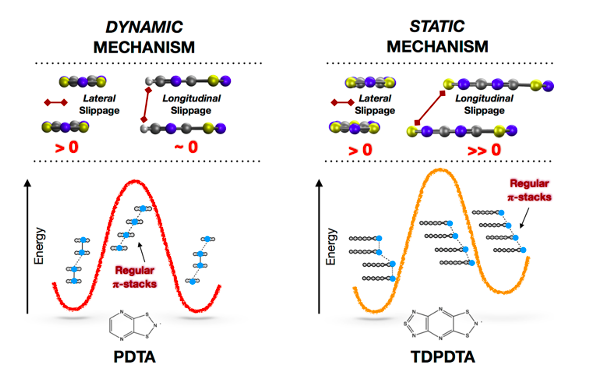
Materials based on regular π-stacks of planar organic radicals are intensively pursued by virtue of their technologically relevant properties. Yet, these π-stacks are commonly unstable against π-dimerization. In this computational study, we reveal that regular π-stacks of planar dithiazolyl radicals can be rendered stable, in some range of temperatures, via two different mechanisms. When the radicals of a π-stack are both longitudinally and latitudinally slipped with respect to each other, the corresponding regular π-stacked configuration is associated with a locally stable minimum in the potential energy surface of the system. Conversely, those regular π-stacks in which radicals are latitudinally slipped with respect to each other are stable as a result of a dynamic interconversion between two degenerate dimerized configurations. The existence of two stabilization mechanisms, which can be traced back to the bonding properties of isolated π-dimers, translates into two different ways of exploiting spin-Peierls-like transitions in switchable dithiazolyl-based materials.
