Reorganization of intermolecular interactions in the polymorphic phase transition of a prototypical dithiazolyl-based bistable material
T. Francese, F. Mota, M. Deumal, J. J. Novoa, R. W. A. Havenith, R. Broer, J. Ribas- Arino.
Cryst. Growth Des., 19 (2019) 2329.
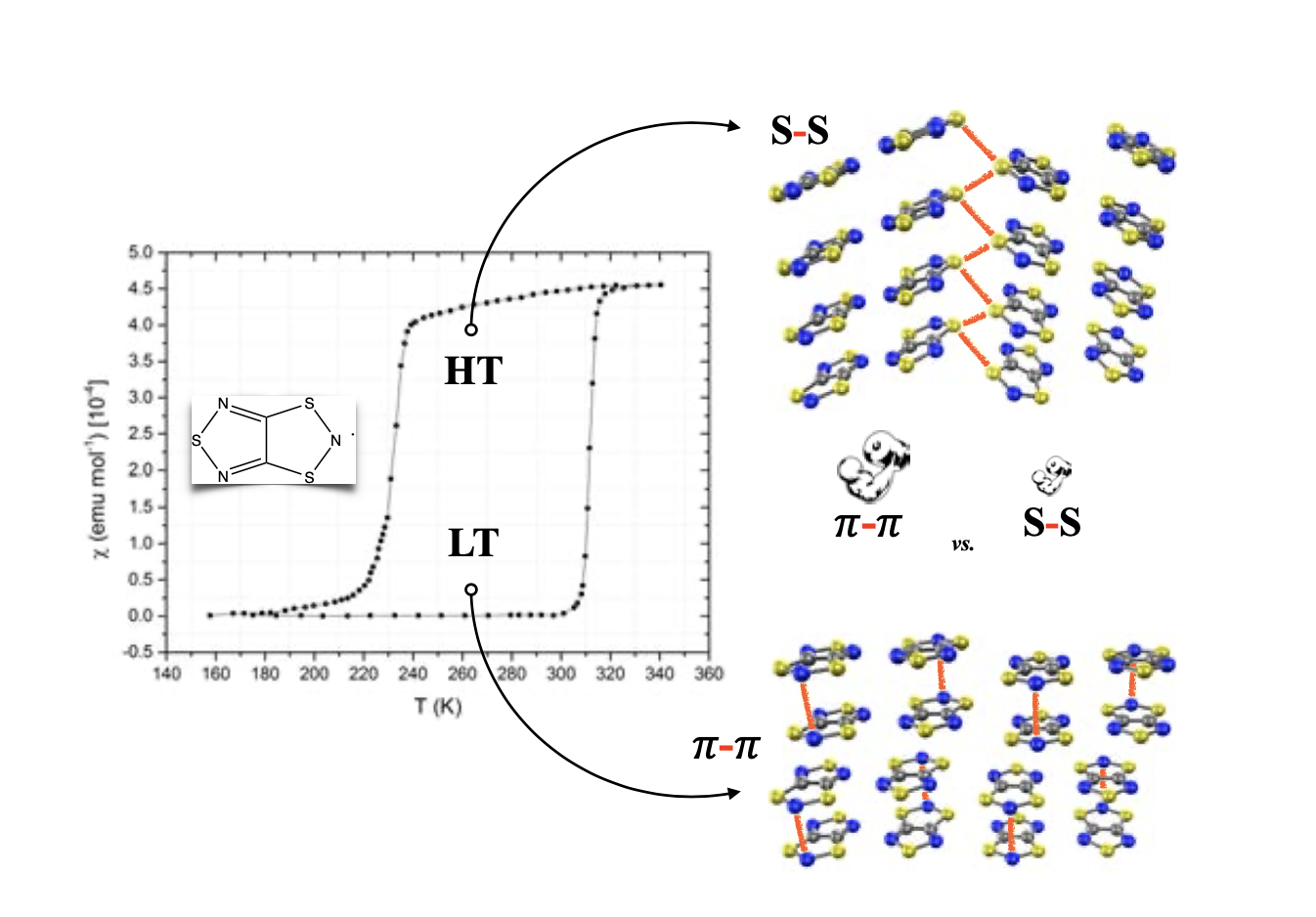
The spin transitions undergone by several molecularcrystals of dithiazolyl (DTA) radicals make this type of radical promisingcandidates for future sensors and memory devices. In this work, we performed asystematic computational study of the intermolecular interactions existingin the two polymorphs of the neutral radical 1,3,5-trithia-2,4,6-triazapentalenyl in order to elucidate the origin of the difference inenergy between those two polymorphs involved in its spin transition andto understand the crystal packing of this prototype of bistable materials.The π−π interactions between radicals are the main driving force for thecrystal packing of both polymorphs, which comprises π-stacks of radicals. The difference in energy betweenpolymorphs, in turn, is mainly controlled also by the intrastack π−πintermolecular interactions, and the interstack S···S contacts. Since the supramolecular motifs herein identified as important for the crystal packing and/or for the energy difference between polymorphs (and, thus, for the spin transition temperature) are common to other members of the DTA family, our results provide valuable information to understand better the structure and properties of other switchable DTA- based materials.
