Tuning Single-Molecule Conductance in Metalloporphyrin-Based Wires via Supramolecular Interactions.
A. C. Aragonès, A. Martin-Rodríguez, D. Aravena, J. Puigmartí-Luis, D. B. Amabilino, N. Aliaga-Alcalde, A. Gonzalez-Campo, E. Ruiz, I. Díez-Pérez. Angew. Chem. Int. Ed., 59 (2020) 19193-19201.
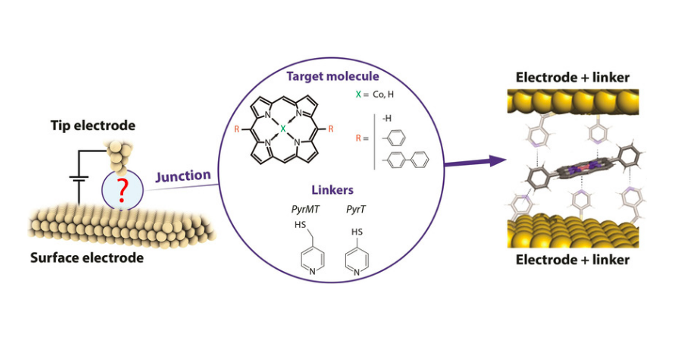
Nature has developed supramolecular constructs to deliver outstanding charge-transport capabilities using metalloporphyrin-based supramolecular arrays. Herein we incorporate simple, naturally inspired supramolecular interactions via the axial complexation of metalloporphyrins into the formation of a single-molecule wire in a nanoscale gap. Small structural changes in the axial coordinating linkers result in dramatic changes in the transport properties of the metalloporphyrin-based wire. The increased flexibility of a pyridine-4-yl-methanethiol ligand due to an extra methyl group, as compared to a more rigid 4-pyridinethiol linker, allows the pyridine-4-yl-methanethiol ligand to adopt an unexpected highly conductive stacked structure between the two junction electrodes and the metalloporphyrin ring. DFT calculations reveal a molecular junction structure composed of a shifted stack of the two pyridinic linkers and the metalloporphyrin ring. In contrast, the more rigid 4-mercaptopyridine ligand presents a more classical lifted octahedral coordination of the metalloporphyrin metal center, leading to a longer electron pathway of lower conductance.
